
Drug Eluting Balloon Market Size, Growth, Trends and Forecast to 2028 | Medtronic, Becton, Boston Scientific Corporation | Medgadget

Drug-Coated Balloon for De Novo Coronary Artery Disease: JACC State-of-the-Art Review | Journal of the American College of Cardiology

Medtronic delivers 5-year data for its drug-coated balloon for PAD, compared to drugless angioplasty | Fierce Biotech

New MEDTRONIC ADM05004008P In.Pact Admiral Paclitaxel-Coated PTA Balloon Catheter, 5.0mm x 40mm, 80cm (X) Disposables - General For Sale - DOTmed Listing #3519528:

Long-Term Clinical Effectiveness of a Drug-Coated Balloon for the Treatment of Femoropopliteal Lesions | Circulation: Cardiovascular Interventions

Medtronic to manufacture IN.PACT Admiral drug-coated balloon at new high-tech facility - Vascular News








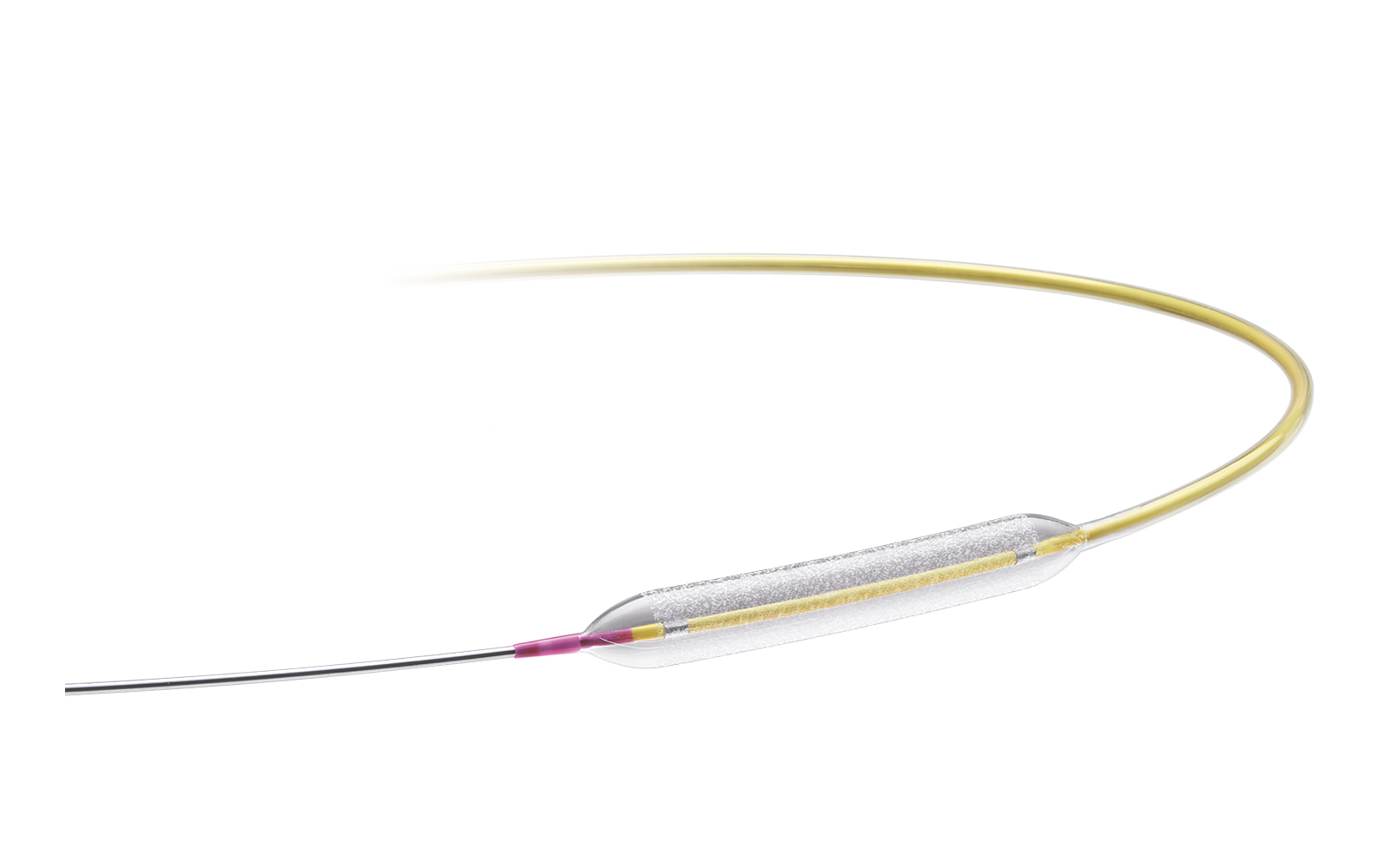


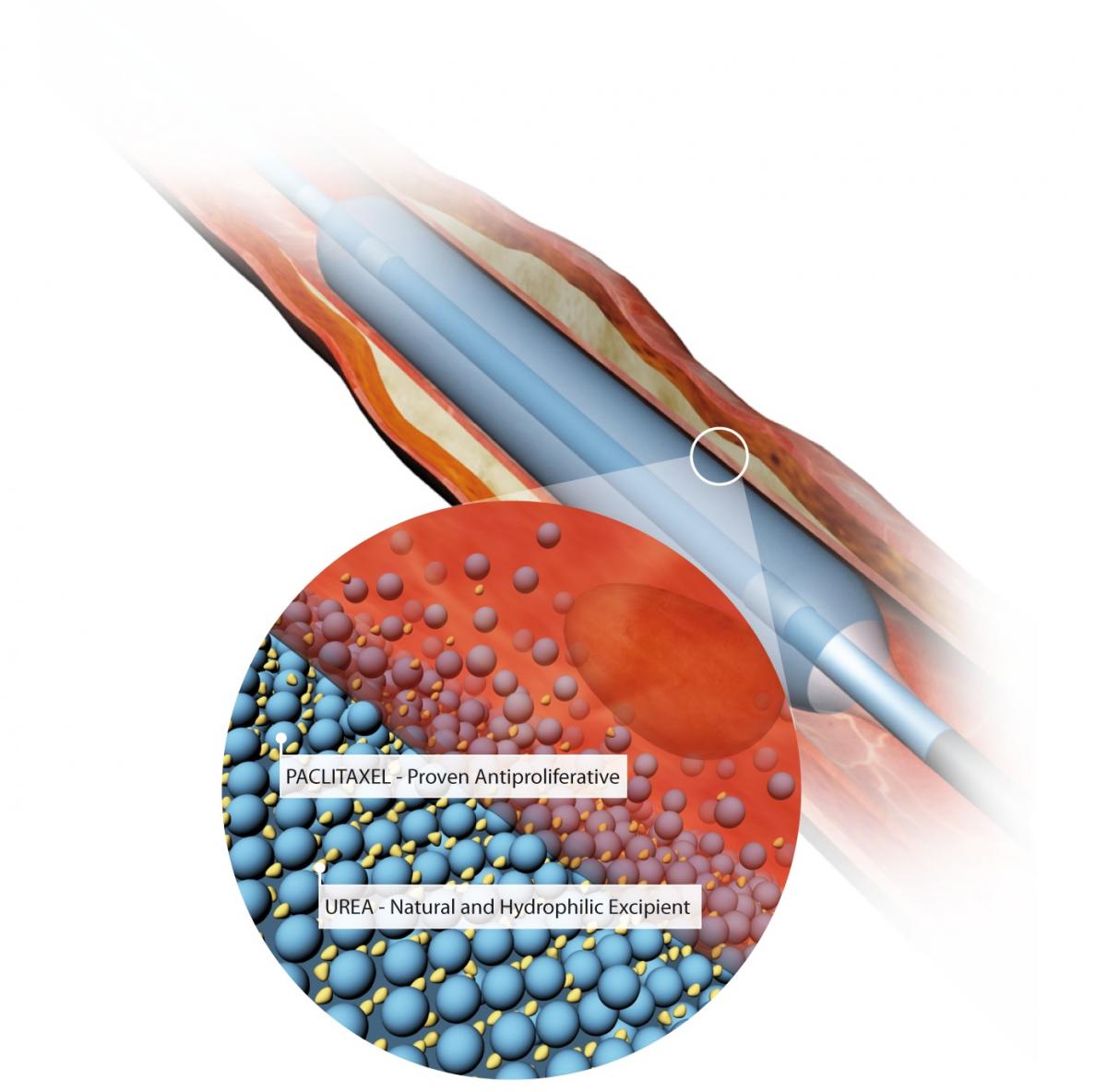


_1653668541.jpg)
