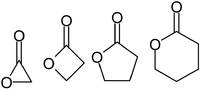
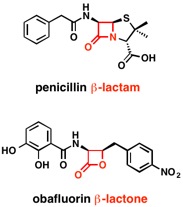
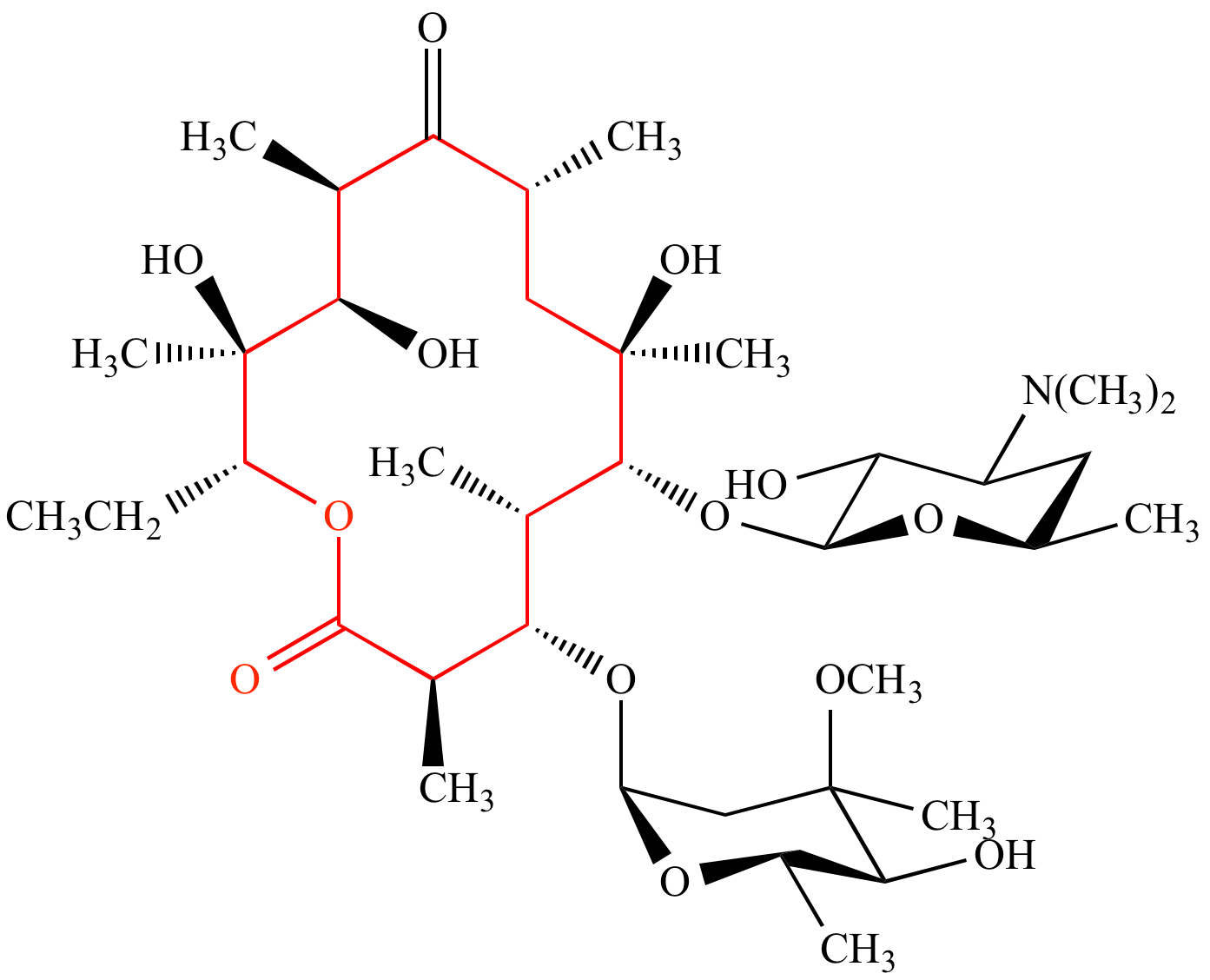
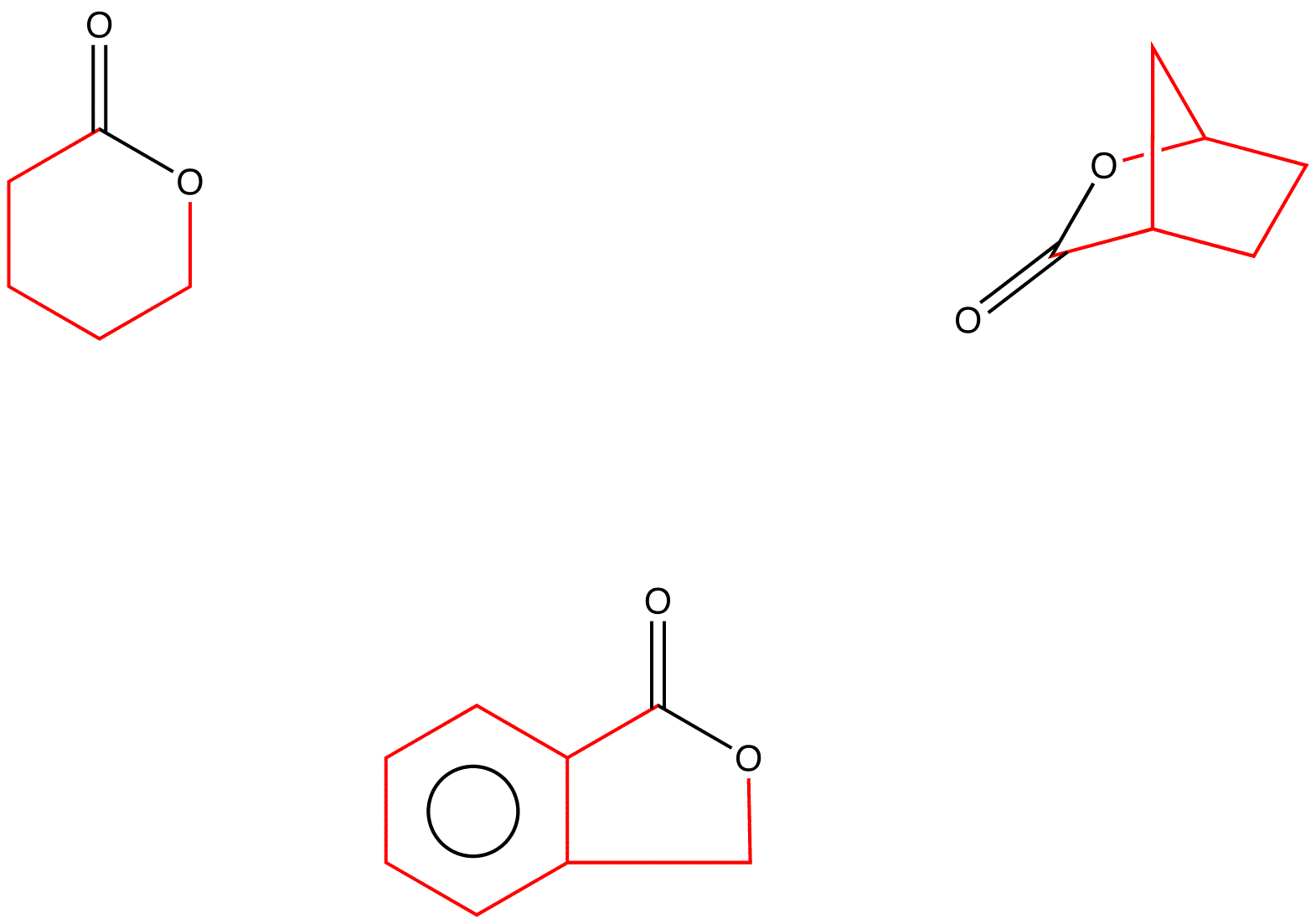
Lactones: Classification, synthesis, biological activities, and industrial applications - ScienceDirect
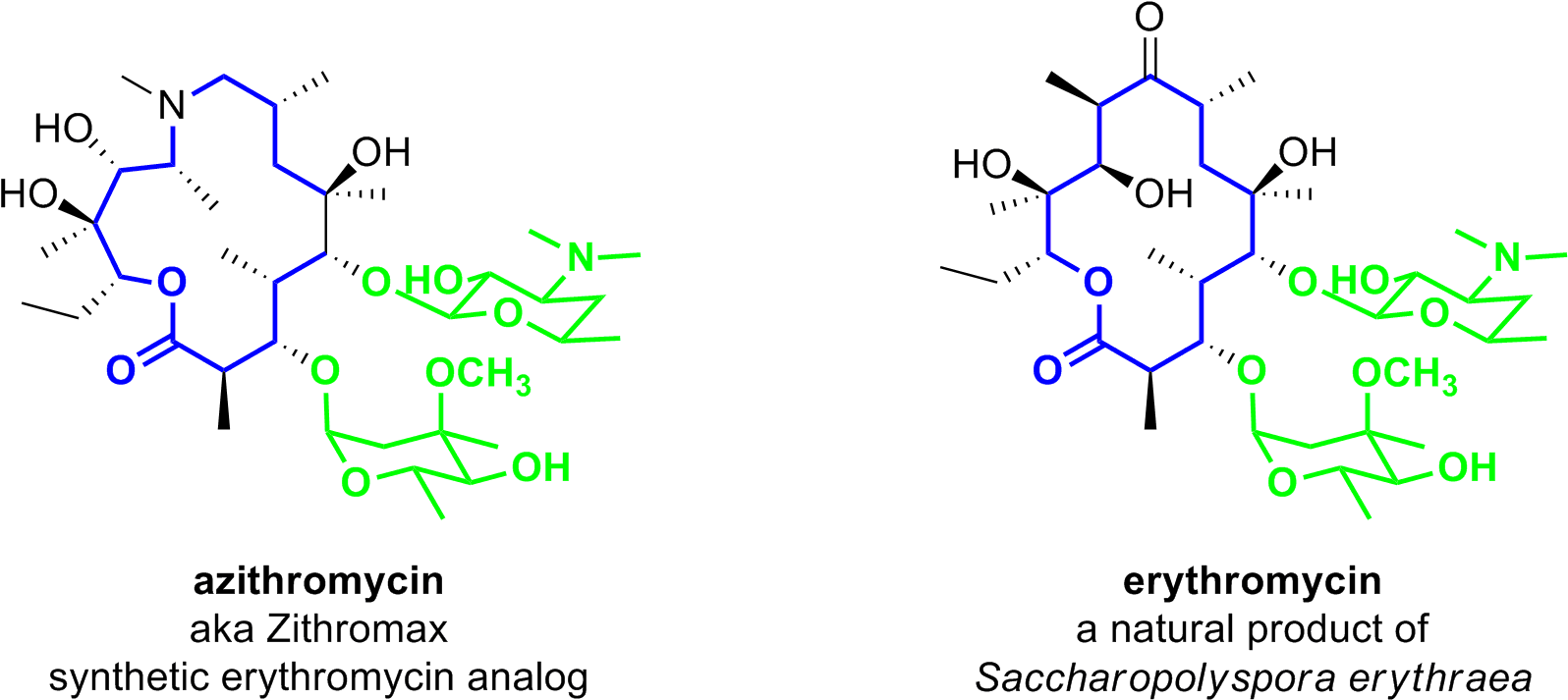
Download Macrolide-antibiotics - Lactone Ring In Azithromycin PNG Image with No Background - PNGkey.com

Controlled Random Terpolymerization of β-Propiolactone, Epoxides, and CO2 via Regioselective Lactone Ring Opening | CCS Chem
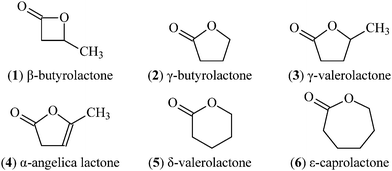
Degradable polymers from ring-opening polymerization of α-angelica lactone , a five-membered unsaturated lactone - Polymer Chemistry (RSC Publishing) DOI:10.1039/C1PY00067E

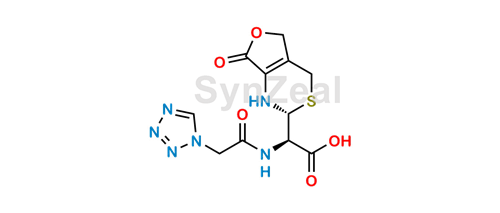
Mild and Chemoselective Lactone Ring-Opening with (TMS)ONa. Mechanistic Studies and Application to Sweroside Derivatives | The Journal of Organic Chemistry

Mild and Chemoselective Lactone Ring-Opening with (TMS)ONa. Mechanistic Studies and Application to Sweroside Derivatives | The Journal of Organic Chemistry

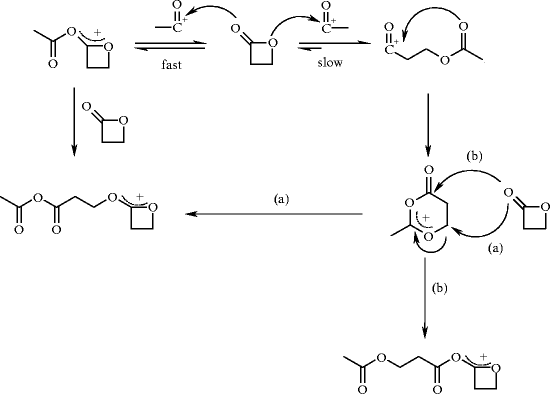
Organocatalysis applied to the ring‐opening polymerization of β‐lactones: A brief overview - Khalil - 2019 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library

SciELO - Brasil - Synthesis of medium ring and macrocyclic acetylenic lactones by the ring expansion of oxabicycloalkenones Synthesis of medium ring and macrocyclic acetylenic lactones by the ring expansion of oxabicycloalkenones

Organocatalysis applied to the ring‐opening polymerization of β‐lactones: A brief overview - Khalil - 2019 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library

Biosynthesis of ebelactone A: isotopic tracer, advanced precursor and genetic studies reveal a thioesterase-independent cyclization to give a polyketide β-lactone | The Journal of Antibiotics
Structural Basis of the γ-Lactone-Ring Formation in Ascorbic Acid Biosynthesis by the Senescence Marker Protein-30/Gluconolactonase | PLOS ONE

Organocatalysis applied to the ring‐opening polymerization of β‐lactones: A brief overview - Khalil - 2019 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library