FDA approves GSK's Nucala for use in adults with chronic rhinosinusitis with nasal polyps – PharmaLive
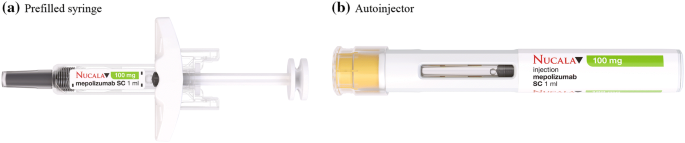
Mepolizumab prefilled syringe and autoinjector: a profile of their use in severe eosinophilic asthma | SpringerLink
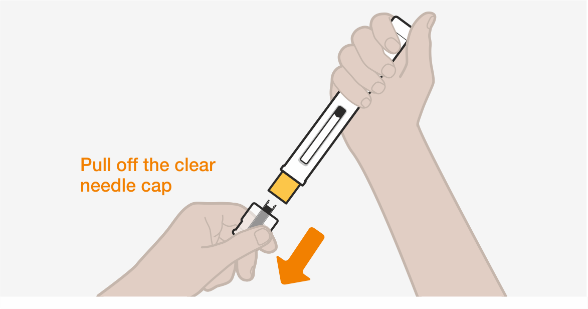
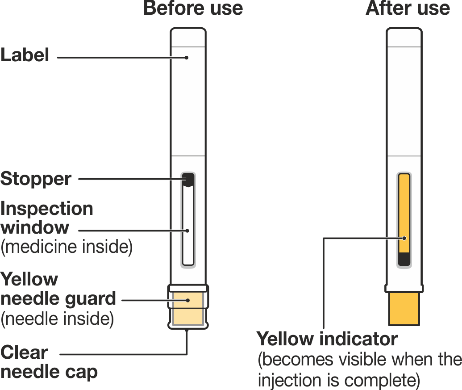
INSTRUCTIONS FOR USE NUCALA (mepolizumab) solution for injection in pre-filled pen (auto-injector) Administer once every 4 weeks